What is soil pH? Soil pH measures the concentration of Hydrogen ions (H+) in the soil. Almost every farmer is familiar with looking at the soil pH on their tests. However, often this is viewed as a secondary concern. The main concern to the soil is the availability of Phosphorous, Potassium, and Nitrogen to the plant. Soil pH tends to take a back seat a lot of the time. However, if this is your viewpoint of this soil then you need to keep reading because what this article is going to demonstrate is not only that pH should be the first thing looked at on a soil report but also how everything else we are building our growth programs on relies on that pH. Proper soil pH is the foundation of everything we build on in crop fertility.
To discuss the importance of proper soil pH, we first need to establish what pH is and what the desired levels are in the soil. The acronym pH stands for Potential Hydrogen. Essentially, we are looking at the amount of hydrogen in the ground. This is very similar to looking at a glass of water, but rather than measuring how much water is in the glass. We measure how much air is in the glass, and from that, we can deduce that the remainder of the glass should be filled with water, giving us an understanding of how much water we need in order to fill the glass. For most crops, the desired pH is anywhere from 6.0 – 7.0 for proper plant growth but for optimal soil performance in most crops, we shoot for a soil pH of 6.7 to 6.9. This provides a glass that is mostly full but still allows for some space that is healthy for soil performance.
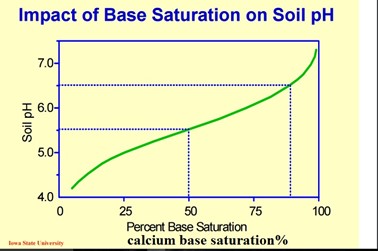
In the soil, we use pH to understand the distribution of soil Cations. Healthy soil will typically be made up of 75% Ca (Calcium), 12-15% Mg (magnesium), 3-5% K (Potassium), 10% or less H+ (Hydron or Hydrogen), and less than 1% Na (sodium).
Now for the main question at hand, why is this so important? When it comes to soil health, having the proper nutrients in the soil determines many things about soil health such as proper pH determines the availability of almost all essential plant nutrients, pH is crucial in weed control, pH directly affects the presence of positive bacteria in the soil, water distribution, compaction control, root development, drought resistance, and so much more. When the statement is bidding that pH is the foundation of everything your crop is built on, it really is almost EVERYTHING.
Calcium and its Role in pH
As stated previously, pH is, more than anything, a way of measuring whether there are enough nutrients in the ground to maintain proper soil health. Calcium makes up 75% of these needed nutrients. It goes without saying just how important calcium is to the ground. Calcium is what we call a trucker nutrient, which means it is directly responsible for moving needed plant nutrients through the soil and into the plant. But calcium is not important only to the soil but the plant as well. Calcium makes up the cell wall of the plant. Just like our bones are made of calcium, the same goes for plants. Without proper calcium levels, this can hinder the plants’ growth and directly impact cell division and reproduction, and a plant’s health and availability to fight disease and drought. In another article, we dive further into the importance of calcium in the soil and in the plant. That article can be found here: (insert article link)
Nutrient Availability
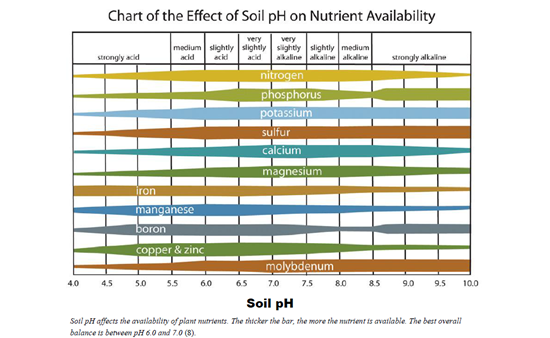
Many Universities have conducted soil studies showing the direct impact of soil pH on nutrient availability. In this article, we are going to focus on research conducted by Iowa State University.
Figure 2 demonstrates the direct correlation between nutrient availability in the soil and pH. The more acidic the soil becomes, the more the nutrients in the soil become bound up. This not only can adversely substantially impact crop product and yield but can also have a direct effect on the availability of the fertilizers that we are applying to the soil. In a time when fertilizer is significantly more expensive than it has ever been, this can mean massive potential yield losses and unnecessary additional financial input. By first fixing pH before fertilizing, we can not only help to increase yield but significantly help lower our input costs as well to achieve the same or greater production goals. Figure 3 demonstrates just how much of an impact the soil pH can have on applied fertilizer.

Weed Control
Every farmer knows the pain of fighting weeds and the yearly applications of herbicides to try and control weed presence in the soil. When it comes to weed control, a lot of the weeds that we, as farmers, fight on a yearly basis are a direct reflection of soil health. The unhealthier the soil becomes and the lower the pH gets, the easier it makes it for many of those weeds to infiltrate and take over the soil. This is especially difficult in hay and pasture situations when dealing with weeds such as broomsedge. By increasing soil health and raising pH, we can not only increase our crop’s performance but help to drive out those weeds hurting our production the most.
Soil pH and Soil Conditioning
Another important aspect of soil pH and proper nutrient distribution in the ground has to do with soil conditioning. In low-pH soil, the ground can become increasingly tight. This restricts water absorption, nutrient movement, soil microbial life, and root development. On the other hand, when magnesium levels are too high in the soil, the soil can become very stick, causing unhealthy water retention, nutrient immobilization, and more. Calcium is what is known as a natural soil aerator. By ensuring the proper calcium levels in the soil and, therefore, the proper pH, we can ensure that we are setting up our soil to regulate water, encourage positive bacteria in the ground, increase nutrient mobilization, improve root development, and so much more.
The importance of soil pH cannot be overstated. The next time you are looking at your soil test, take a moment, look at your soil pH, and look at your base saturation. Ensure that you have the right foundation to build your fertilizer program on and, therefore, your crop.
